Causes of Skin Cancer: Genetic Mutations
Reviewed by: HU Medical Review Board | Last reviewed: May 2017. | Last updated: October 2021
The US Department of Health and Human Services lists ultraviolet (UV) radiation as a carcinogen or cancer-causing substance.1 Sunlight and indoor tanning beds produce UV light. UV rays can damage the DNA in skin cells. Harmful DNA changes, called genetic mutations, can lead to skin cancer and uncontrolled cell growth.
UV light exposure is one of the most important environmental risk factors for most forms of skin cancer. By limiting UV exposure, you can reduce your chance of getting skin cancer.
Why is it important to understand genetic mutations in skin cancer?
It is helpful to know about genetic mutations in order to understand how certain medications work. Some of the newer medications for skin cancer are called targeted therapies. These medications interfere with molecular “targets” that have a role in cancer cell growth and survival.2,3 Cancer cells are different from normal cells. Targeted therapies aim at certain features that make the cancer cells different.
There are several targeted therapies approved for skin cancer. In general, these drugs block or interfere with certain areas of cancer cells that cause a cancer cell to grow and divide.
Types of genetic mutations
Some of the significant genetic changes in basal cell carcinoma (BCC) affect a cell signaling pathway called Sonic Hedgehog. In particular, some of these genes that may carry mutations include:
- PTCH1
- SMO
Genetic mutations have a role in melanoma too. Some of these genes that may carry mutations in melanoma are named:4
- BRAF
- NRAS
One of the most common genetic mutations in squamous cell carcinoma involves the TP53 gene.5
What is the Sonic Hedgehog pathway?
Some medications used to treat advanced basal cell carcinoma affect a cell signaling pathway called Sonic Hedgehog. (Yes, like the video game.)6
Before you were born, the Sonic Hedgehog pathway had an important role in the development of your organs and making sure the right side of your body matches your left. This pathway is not active in most healthy adult tissue, but skin is an exception.7 In skin, Sonic Hedgehog maintains the supply of stem cells. Stem cells are flexible cells that can develop into many different cell types. The Sonic Hedgehog pathway has a role in the development of your hair follicles and glands and it helps to control skin growth.
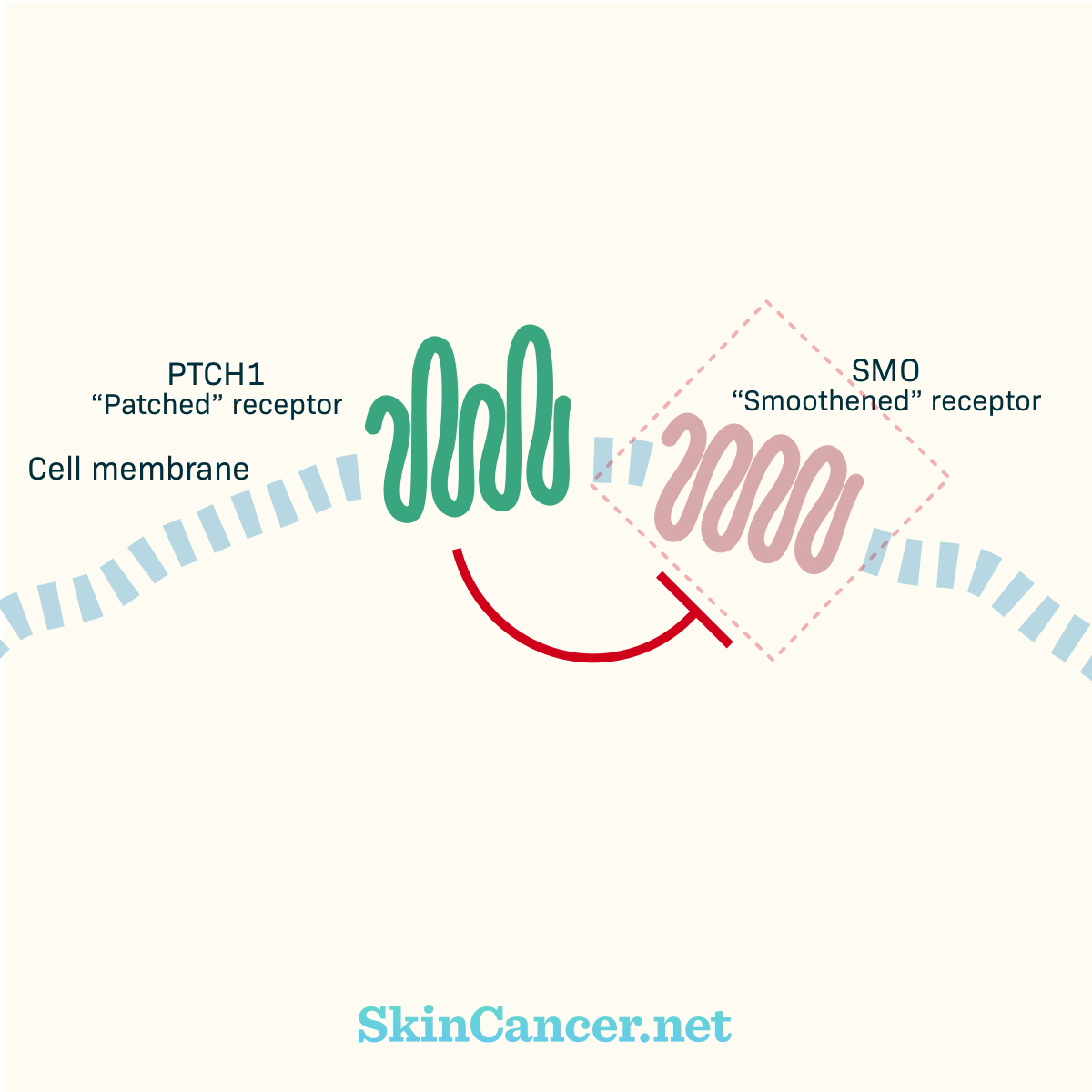
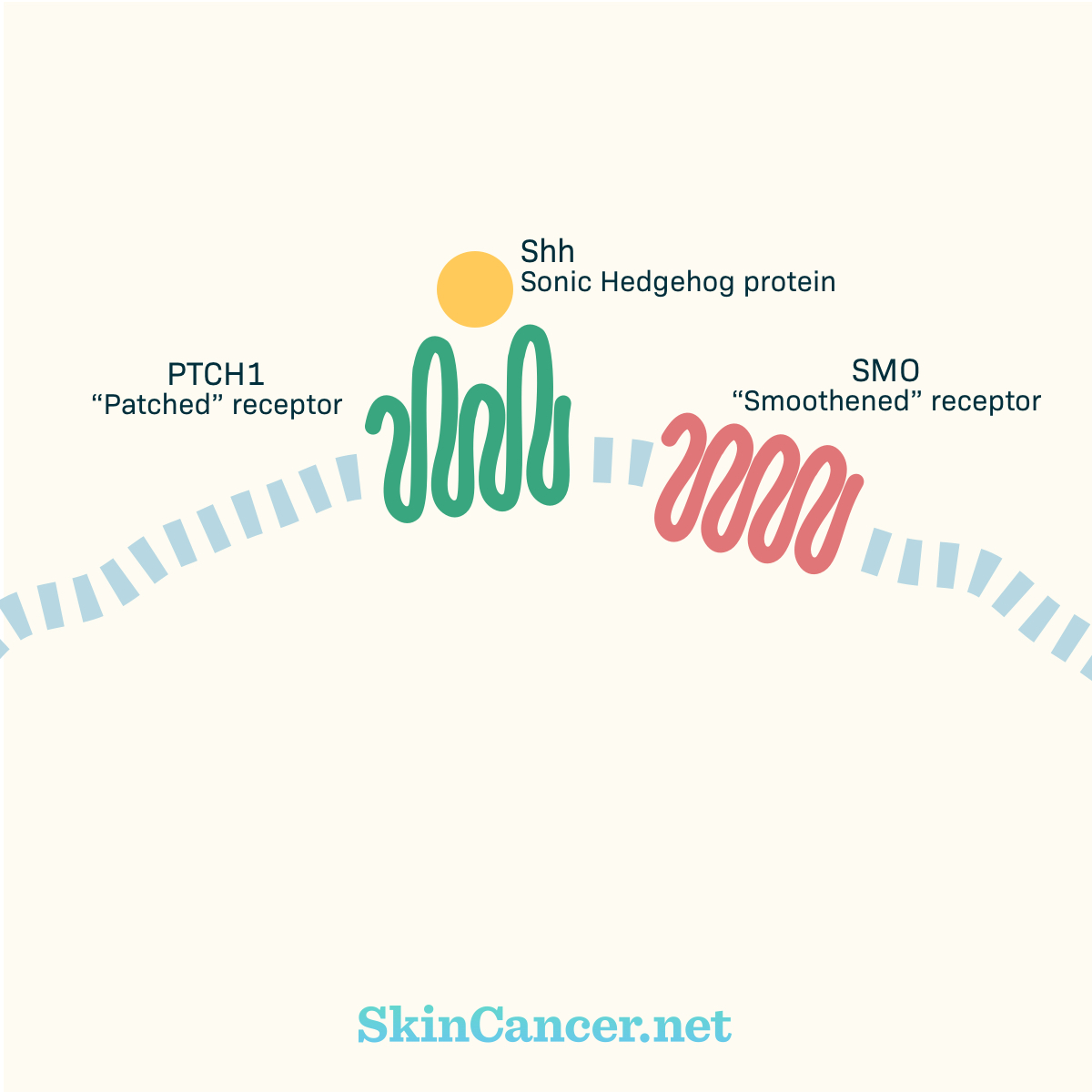
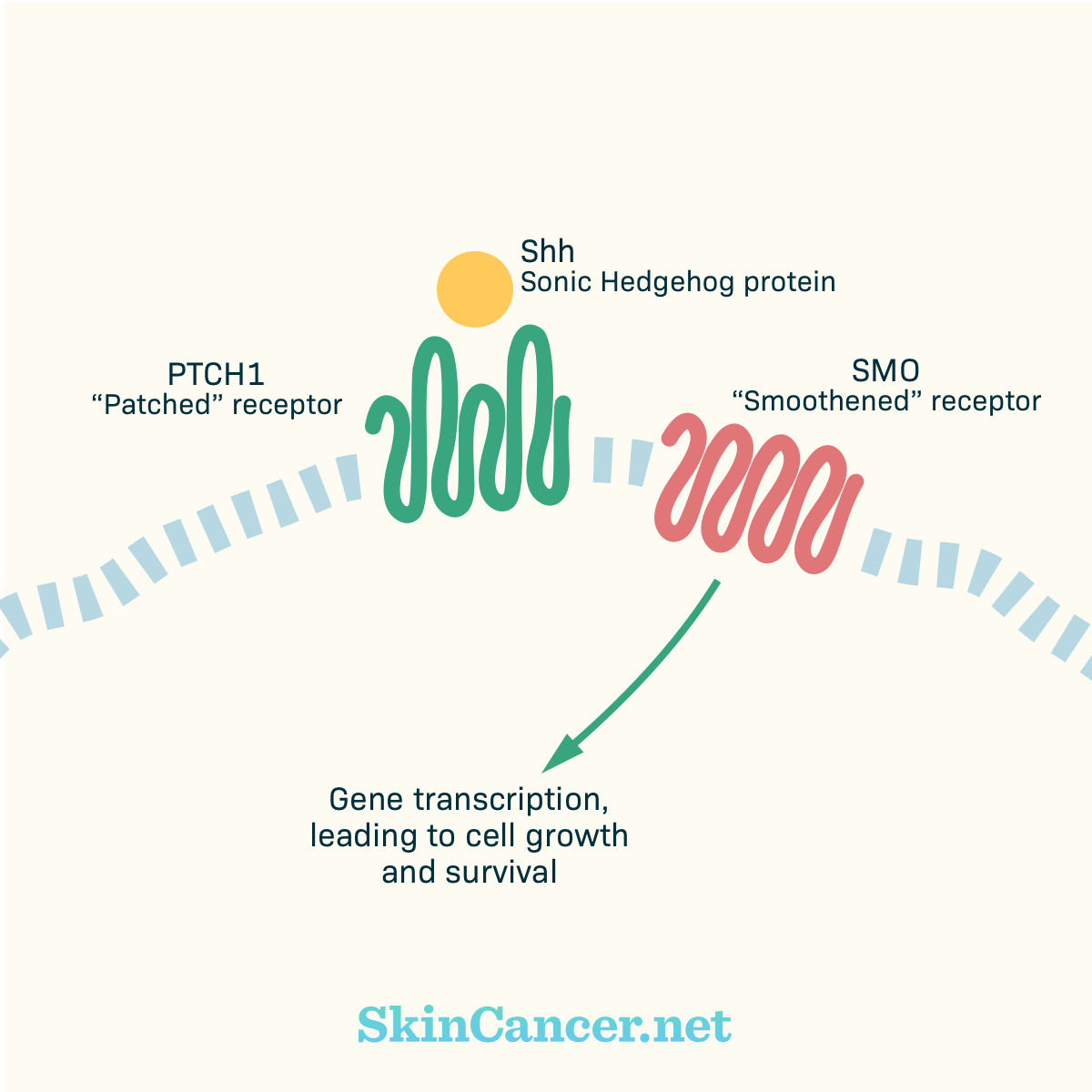
Key players in the Sonic Hedgehog pathway are two receptors called PTCH1 (“Patched”) and SMO (“Smoothened”).7 Figure 1 illustrates how the Sonic Hedgehog pathway works. (Please note: This is a simplified explanation. Although some details are missing, it illustrates the basic way that the receptors interact with each other.) You can imagine that PTCH1 works like a door, holding SMO in a little box. The Sonic Hedgehog protein is a key to open the box (Figure 2). When Sonic Hedgehog protein opens up the PTCH1 door, SMO is free to start working (Figure 3). It starts sending signals that cause cells to grow and survive.
Figure 1. PTCH1 and SMO signaling
PTCH1 is a receptor in the cell membrane. Without the Sonic Hedgehog protein (SHH), PTCH1 blocks SMO, another receptor, from sending any signals. When the SHH protein binds to PTCH1, the SMO travels to the cell surface. SMO begins sending signals that lead to cell growth and survival.
Figure 2. Sonic Hedgehog protein binding
PTCH1 is known as a “tumor suppressor gene.” By keeping SMO inactive, PTCH1 acts as an “off switch” for the process that causes cells to divide and survive.5 But PTCH1 is mutated in about 60% of BCC tumors.8 When PTCH1 becomes mutated, the “off switch” no longer works, and SMO signals freely. The result is uncontrolled cellular growth. Two medications, Erivedge® (vismodegib) and Odomzo® (sonidegib), work by blocking the SMO receptor.
Figure 3. SMO mutation
SMO is known as an “proto-oncogene” or a type of gene that causes cells to grow, divide, and stay alive.5,9 Certain mutations to SMO cause it to stay “on” all the time, leading to cancer development. SMO is mutated in 10-20% of BCC.15 When SMO is mutated, the medications that block SMO may not work.10
What genetic changes lead to melanoma?
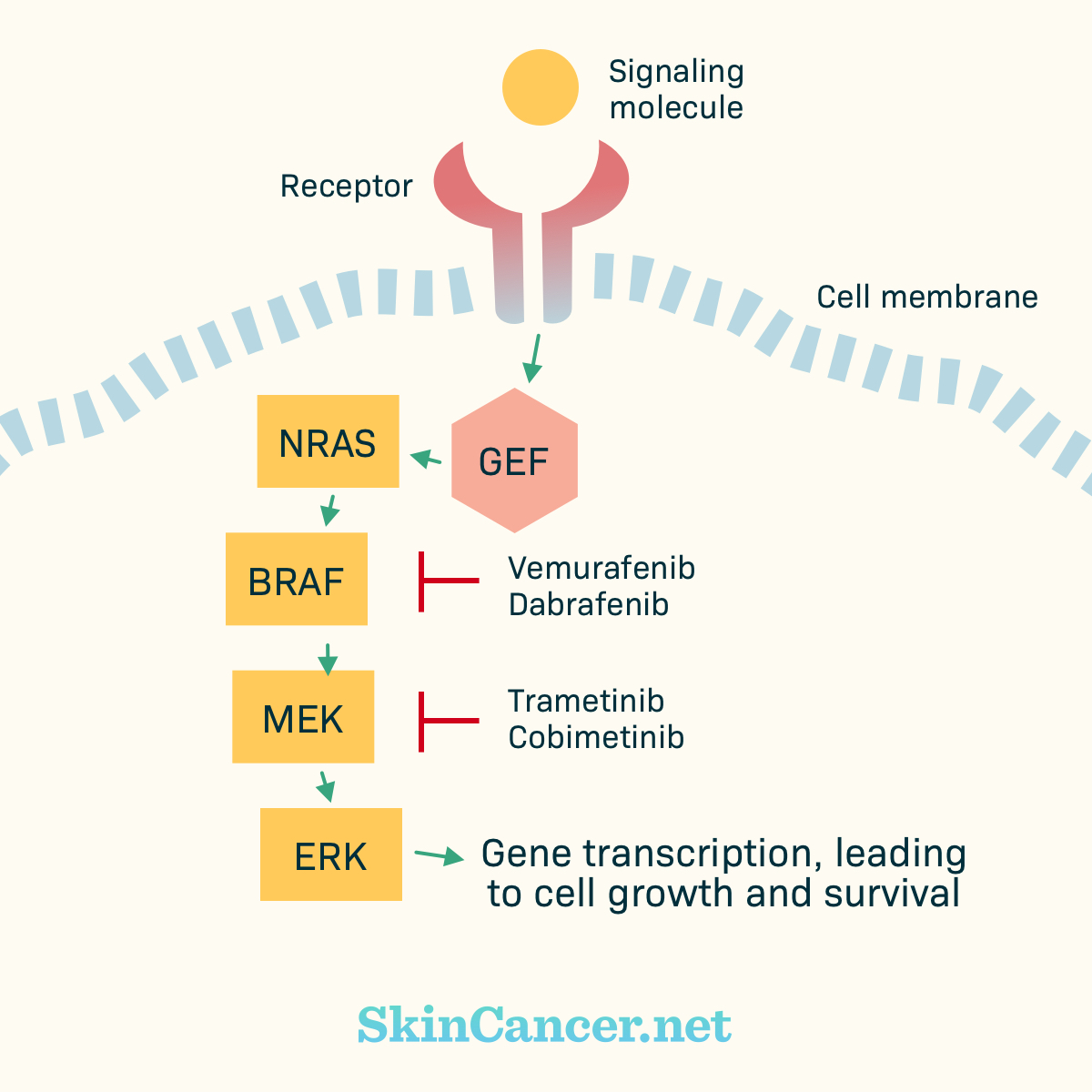
Some of the most common mutations in melanoma are in genes called BRAF and NRAS.4 BRAF and NRAS proteins have a role in a pathway called MAPK or ERK.11 These proteins are part of a chain of events that allow cells to grow and survive. Normally, there are mechanisms that turn each protein “on” and “off,” keeping the cell processes under control.
Figure 4. Simplified MAPK Pathway
Certain mutations cause BRAF to stay “on,” sending continuous signals for cell growth.12 Two medications used to treat melanoma, Zelboraf® (vemurafenib), Tafinlar® (dabrafenib),and BRAFTOVI® (encorafenib) help to block the BRAF protein. Certain BRAF inhibitors may be used together with specific medications that block MEK, which is another protein in the chain of events. Three MEK inhibitors used for melanoma are Mekinist® (trametinib), Cotellic® (cobimetinib), and Mektovi® (binimetinib).
What other mutations are common
TP53 is a tumor suppressor gene that is mutated in about 50% of BCC tumors and up to 90% of squamous cell carcinoma tumors.15,13 Inherited mutations in other tumor suppressor genes called CDKN2A and CDK4 have been seen in melanoma that runs in families.14 These mutations may be future targets for therapy.